Lifelines
BEST EFFORT
Lifelines
BEST EFFORT
Upper airway lifelines
When successful, the three upper airway lifelines of face mask (FMV), supraglottic airway (SGA) and endotracheal tube (ETT) are equally able to fulfil the goal of alveolar oxygen delivery but differ in their ability to fulfil secondary goals such as airway protection, airway security, and carbon dioxide elimination. Whilst important, these secondary goals become inconsequential if alveolar oxygen delivery cannot be achieved.
In routine circumstances, achieving alveolar oxygen delivery simply involves using a lifeline which has been selected to satisfy both the primary and secondary airway management goals necessitated by the clinical situation. When airway management becomes challenging, however, secondary goals may have to be compromised to ensure alveolar oxygen delivery is achieved.
The Vortex Approach allows clinicians the flexibility to initiate airway management using whichever lifeline is felt to be most appropriate to the clinical situation. If initial attempts to establish alveolar oxygen delivery are challenging, the remaining lifelines can also be implemented in whatever sequence is deemed most appropriate in the clinical circumstances. This flexibility in the sequence in which lifelines are attempted allows the Vortex to be applied to any context in which airway management occurs.
BEST EFFORTS
The term 'best effort' is used by the Vortex Approach to describe the circumstance in which all viable strategies to facilitate success at entering the Green Zone via a given lifeline have been implemented. Up to three attempts (+/- a 'gamechanger' - see below), each incorporating additional optimisations that have not previously been implemented, are permitted to complete a best effort. The specific optimisations employed and the number of attempts that are appropriate to achieve a best effort are context dependent decisions, to be made by the airway operator within the confines of the principles set out by the Vortex Approach.
If following a completed best effort at a particular lifeline, alveolar oxygen delivery has not been achieved, then no further attempts at that lifeline should occur. Efforts should instead focus on establishing alveolar oxygen delivery via an alternate lifeline - or on initiating Neck Rescue if all three lifelines have been unsuccessful.
optimisations
The Vortex model prompts five categories of optimisation that may be applied to improve success entering the Green Zone via any of the lifelines.
These five categories apply equally to each of the three lifelines. The specific interventions within each category are discussed elsewhere in relation to the individual lifelines. Categorising optimisations in this manner allows the entire team to track what interventions have been implemented by the airway operator and to offer suggestions in a structured way.
It is not intended that all the optimisation interventions in a given category are exhaustively implemented for a particular lifeline - this would be both time consuming and inappropriate in most circumstances. Instead the optimisation headings serve to encourage the clinical team to consider all of the options, with the airway operator only implementing those thought to be beneficial in a particular context. This structured approach to considering optimisation strategies maximises the opportunities for achieving timely entry to the Green Zone by ensuring that the process of achieving a best effort is:
Efficient: by minimising both the time and number of attempts to implement all strategies considered to be useful.
Rigorous: by minimising the likelihood that potentially helpful interventions are overlooked
Defined: by outlining a discrete set of optimisations for a given lifeline and thereby providing an endpoint to optimisation. This promotes team recognition that a best effort at a given lifeline has been completed and the need to move on to an alternate technique if alveolar oxygen delivery has not been achieved.
The goal is to maximise opportunities to enter into the Green Zone in the shortest possible time. This makes optimal use of the safe apnoea time and minimises the risk that the patient will be exposed to critical hypoxia.
Note that the optimisation prompts listed on the Vortex tool focus on strategies that can be implemented in real time during management of the challenging airway. As such there are additional optimisations (e.g. shaving a beard) that are not prompted by the tool which can be incorporated prior to initiating airway management. The following "training matrix" gives an overview of the real-time optimisations that can be implemented for each of the upper airway lifelines. It is not intended as an implementation tool for use during an airway crisis. The impact of each of these interventions (& thus the difficulties with establishing an airway with each of the lifelines that provide the indication for their use) are dealt with in more detail below.
Attempts
Except in extraordinary circumstances, where it is considered that a lifeline has a negligible chance of success in a particular clinical situation (particularly if the patient is already severely hypoxic), at least one attempt at each lifeline is usually indicated prior to initiating CICO Rescue. Although desirable, it is not usually possible to implement all optimisations required to maximise success at a given lifeline and achieve a best effort on the first attempt. Additional factors which might improve the chances of entering the Green Zone may only be identified after initial airway manipulations have taken place.
The Vortex model allows up to three attempts to achieve a best effort at each lifeline but emphasises using the minimum number of attempts required. Limiting the number of attempts at each lifeline is important from two perspectives:
Trauma: repeated instrumentation of the airway has the potential to cause trauma which can compromise subsequent success with any of the lifelines and convert what would otherwise have been a "can oxygenate" situation into a "can't oxygenate" situation. Repeated airway instrumentation in desperation is also known to have caused additional trauma resulting in patient morbidity beyond airway compromise.
Time: repeated airway instrumentation also consumes time, delaying implementation of alternate techniques and potentially increasing the duration and severity of hypoxaemia to which the patient may be exposed before entry to the Green Zone is established.
The definition of an attempt varies according to the lifeline being implemented.
For ETT it is defined as the insertion and removal of laryngoscope from the airway.
For SGA it is defined as the insertion and removal of a supraglottic airway from the airway.
For FMV it is defined as the application and removal of a face mask to the patient's face in association with intervening generation of positive pressure with the intent of achieving alveolar ventilation
It is not necessary that all attempts in pursuit of a best effort at one lifeline must be completed before initiating the first attempt at an alternate lifeline. In keeping with normal clinical practice, best efforts at multiple different lifelines may be proceeding in parallel, with sequential attempts alternating between optimising different lifelines. Thus the Vortex implementation tool tracks 'completed best efforts' at each lifeline rather than the sequence of individual attempts required to achieve them.
CLINICAL INTEGRATION
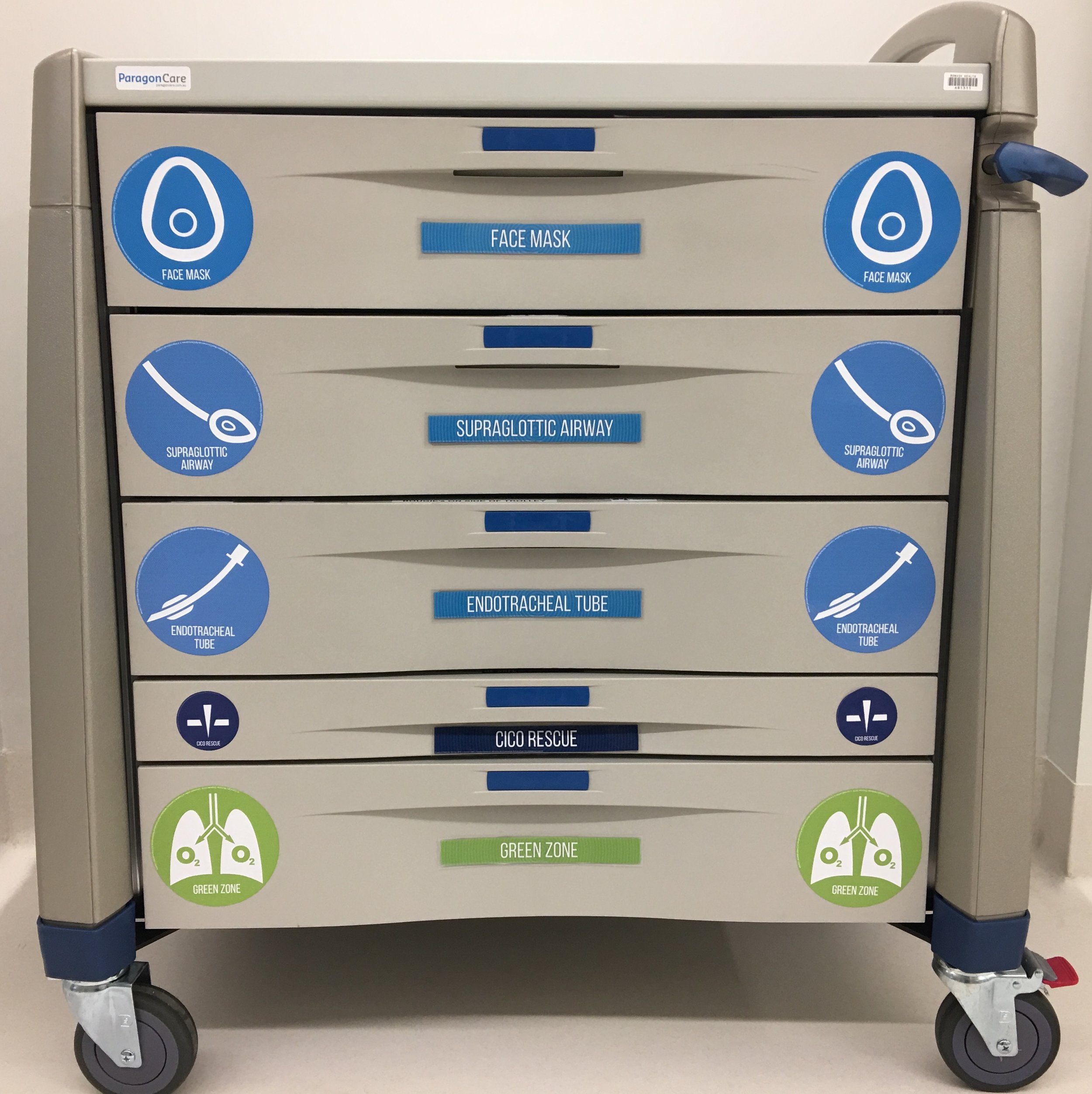
The structured approach to considering optimisation strategies using the five categories of the Vortex for each lifeline can be made even more effective by integrating this approach into the clinical environment. Labelling the emergency airway cart drawers to correspond to the different zones of the Vortex and arranging equipment inside them according to the optimisation categories allows clinicians to use the equipment itself as a prompt to ensure efficient optimisation of the lifelines. Labels to enable this are available for free download.
gamechangers
Using the structured approach provided by the Vortex, it should be possible to implement all the optimisations required to complete a best effort within three attempts at any lifeline. Despite this, it is recognised that circumstances may occasionally exist where a lack of immediate access to personnel, equipment, or medications might prevent crucial optimisations being implemented within the three attempt limit. In this rare situation, the Vortex Approach provides clinicians with permission to have one additional attempt at a lifeline if an optimisation strategy is considered to be a 'gamechanger'.
To be considered a gamechanger an optimisation strategy must satisfy the following criteria:
It must be reasonably expected to have a profound impact on the probability of entering the Green Zone using the lifeline in question: thus an untried intervention that merely provides another possibility of entering the Green Zone is not considered a gamechanger. The specific interventions likely to have a profound impact
It must not prolong the duration or severity of hypoxia to which the patient is exposed: this will require consideration of the patients oxygen saturations, the immediacy with which the proposed optimisation can be implemented and the likelihood of success of any remaining options to establish alveolar oxygen delivery. Thus in the situation where the patient's oxygen saturations have already fallen, unless the clinician is convinced that the proposed optimisation will provide entry to the Green Zone via the relevant lifeline as fast or faster than the remaining alternatives (residual lifelines or Neck Rescue), it cannot be considered a gamechanger.
“a best effort at any lifeline must include full muscle relaxation”
Examples of interventions that might potentially qualify as 'Gamechangers' in appropriate situations, include the following:
Previously unavailable novel equipment that specifically targets the challenges being faced: this includes devices such as a hyperangulated videolaryngoscope or a straight blade direct laryngoscope (in appropriately skilled hands) but not more subtle variations in the size/type of devices which have already used.
Rectification of equipment failure: where equipment failure (e.g. light failure on laryngoscope, screen/camera failure on videolaryngoscope) has prevented proper execution of a previous attempt and the defect can be corrected by device repair/replacement
Full muscle relaxation: a best effort at any lifeline must include full muscle relaxation (unless this cannot be provided within the necessary timeframe) as this will make FMV, SGA, ETT & Neck Rescue easier.
Another clinician of equivalent or increased experience: in addition to technical prowess, the impact of a 'fresh arm' at direct laryngoscopy should not be underestimated as multiple attempts may have resulted in significant fatigue that has counteracted the impact of other optimisation strategies.
An ENT surgeon proficient in rigid laryngoscopy/bronchoscopy and the necessary equipment to perform this procedure.
None of these interventions should be assumed to automatically qualify as gamechangers. The specific interventions likely to satisfy the above criteria will vary according to the clinical situation and the particular challenges faced. The likely impact of a contemplated optimisation strategy on the factor impeding establishment of alveolar oxygen delivery must be considered in context.
A completed best effort at any lifeline may thus consist of up to three attempts +/- a gamechanger. The use of specific, limited criteria which must be satisfied for the gamechanger to be invoked provides boundaries to ensure accountability for this action and avoid fixation on upper airway lifelines when Neck Rescue is clearly indicated.
Human factors
Most of the major difficult airway algorithms emphasise the need to make declarations of "failure" at each of intubation, supraglottic airway and face mask ventilation in order to facilitate team situational awareness of the need to move on to other techniques. Declaring that alveolar oxygen delivery cannot be achieved by a given technique is a key step in encouraging the team to commit to alternate strategies but linking such a declaration to the notion of "failure", with the implications this may carry for the competence of the airway operator, has the potential to become a barrier to such a declaration being made.
Richard Levitan has spoken about the importance of psychology in approaching transition to Neck Rescue and the concept of the "surgically inevitable airway", stressing the need to move away from the perception that the need to perform this procedure indicates inadequacy on the part of the airway clinician. The challenges presented managing any airway are the culmination of patient anatomy, circumstances and clinician factors. While clinician factors certainly include planning and clinical skills (and where these are inadequate they may certainly contribute to the need for Neck Rescue when it could otherwise have been avoided) focusing on this aspect during the transition process is of no conceivable benefit. Faced with an evolving airway crisis the team must do their best within the context they find themselves in. The psychological burden of declaring "failure" has the potential to become an impediment to effective transition even for clinicians who have performed exceptionally and are simply unlucky enough to find themselves presented with the "surgically inevitable airway".
In keeping with this principle, the endpoint of attempts to establish alveolar oxygen delivery via any lifeline using the Vortex Approach is declared by the team as a "completed best effort". This term serves to convey the futility of further attempts at the relevant lifeline while emphasising that the clinician has maximised the the opportunities available to them according to anatomical, situational and clinician factors at the time. The expectation is that there are less barriers to a clinician to declaring a "completed best effort" at intubation than to declaring "failure". If following a completed best effort at any lifeline alveolar oxygen delivery has not been restored, then alternate strategies must be pursued, including Neck Rescue when best efforts at all upper airway lifelines have been exhausted.

Face Mask
BEST EFFORT
Face Mask
BEST EFFORT
Troubleshooting face mask ventilation
Entering the Green Zone using face mask ventilation in an apnoeic patient requires both an effective seal and a patent airway (in a spontaneously ventilating patient only airway patency is needed). Optimisation attempts should be specifically directed towards the the factors impeding alveolar oxygen delivery (recognising that inability to establish a seal and a inability to establish a patent airway may coexist). Targeting optimisations in this way requires clinicians to distinguish between these two issues which may both present as a leak around the face mask during positive pressure ventilation. The leaks resulting from inadequate seal and airway obstruction can be respectively categorised as primary and secondary leaks.
Primary Leaks: these are the direct result of an inadequate face mask seal, allowing gas to escape around the perimeter of the mask, impeding generation of the pressure required to ventilate the alveoli with oxygen (primary leaks may also occur due to equipment faults that allow gas to escape through holes/disconnections in the face mask or other parts of the oxygen delivery system or by failure of gas flow in the absence of a self inflating bag - this latter group of problems is distinguishable by the presence of a primary leak without a discernible leak around the mask and will not be considered further here. The optimisations of the Vortex deal mainly with primary leaks resulting from inadequate mask seal but the potential for issues with equipment issues should always be considered when a primary leak occurs). Primary leaks require either interventions to seal the route by which gas is escaping or provision of sufficient extra gas flow to offset the impact of the gas that is being vented. Interventions to improve airway patency are unlikely to be of assistance.
Secondary Leaks: these occur indirectly due to airway obstruction which is unable to be overcome resulting in elevated inspiratory pressures with positive pressure ventilation. This increased pressure overcomes what would otherwise have been an adequate face mask seal. Thus even with ideal application of a face mask, attempts at positive pressure ventilation of a completely obstructed airway must ultimately lead to escape of gas around the perimeter of the mask when the airway pressure exceeds the seal pressure of the mask. Secondary leaks require interventions to improve airway patency. Interventions to improve the seal around the face mask are less likely to be of assistance unless patency issues have been addressed.
In the presence of both primary and secondary leaks (that are not due to equipment faults) gas escapes around the perimeter of the mask and impaired or absent alveolar ventilation results. The ability to distinguish between these two scenarios is of critical importance however, as it informs the airway operator as to the appropriate optimisation strategies to overcome the problem. Use of a manual ventilation device which has a soft collapsible bag (rather than a self inflating bag) is recommended as it allows these two situations to be easily distinguished.
Primary leaks are identifiable by collapse of the reservoir bag indicating that attention must be directed to optimising face mask seal (notably when these devices are used for preoxygenation, collapse of the reservoir bag often allows these issues to be identified and addressed prior to initiating airway management).
Secondary leaks: in the presence of airway obstruction but adequate primary seal, the soft bag will remain pressurised and inflated, indicating that attention needs to be directed towards optimisations that improve airway patency.
Where primary and secondary leaks coexist, addressing a primary leak may reveal the presence of the secondary leak.
The ability to detect pressure in the manual ventilation bag is thus of critical importance in distinguishing between failures of face mask ventilation due to inadequate seal and those due to airway obstruction. When using manual ventilation devices with a self inflating bag such as a bag-valve-mask (BVM) device, the reservoir bag will remain inflated independent of the ability to pressurise the airway. In addition the stiffer material of the self inflating bag provides limited feedback about the airway pressure being generated during inspiration and significantly limits the ability of the airway operator to identify airway obstruction as the cause of a face mask leak. The consequence is an inability to distinguish between primary and secondary leaks and thus an impaired ability to target face mask optimisations to their underlying cause. The use of a BVM device is thus able to conceal both the presence of a face mask leak and of airway obstruction and may therefore not only impede identification of the cause of inadequate face mask ventilation but delay its recognition entirely. The role of BVM devices is to allow ongoing ventilation in the absence of oxygen flow and while they should always be immediately available for this contingency, devices with self inflating bags cannot be recommended as the primary equipment for ventilation in any other circumstances (one exception is retrieval settings where space/weight requirements may preclude carrying of two separate manual ventilation devices). Devices with a soft reservoir bag such as an anaesthetic circle system or Mapleson circuit are preferred. While devices with a self inflating bag are often perceived by clinicians as being easier to use, this reflects the ability of the self inflating bag to conceal poor technique and inadequate face mask ventilation rather than representing a greater likelihood of success with these devices.
best effort AT FACE MASK VENTILATION
The following table outlines the possible real-time optimisation interventions in pursuit of a best effort at face mask ventilation and the aspects of compromised face mask ventilation they target. It includes only interventions that can be implemented during the process of managing a challenging airway and thus other interventions such as shaving/taping beards which may be useful prior to initiating airway management, have been omitted. Note that this table is intended as a foundation resource for training and not to be used as an implementation tool during the process of airway management.
Note: performance of flextension (sniffing position) must only be undertaken following consideration of the need for cervical spine precautions.
FLEXTENSION:
The term 'flextension' was coined by Prof Tim Cook as a more precise and descriptive term for the 'sniffing position' that specifically emphasises the need for both base-of-neck flexion and atlanto-occiptal extension when performing this manoeuvre. Positioning of the tragus of the ear above the sternal notch with the patient's face parallel with the ceiling is used to provide an objective endpoint for adequate flextension. While 'ear above sternal notch' can be achieved without employing both base of neck flexion and atlanto-occipital extension, this does not have the same beneficial effects on success at each of the upper airway lifelines as achieving this via flextension.
Flextension should only be performed following consideration of the need for cervical spine precautions.
'CLAW' GRIP VERSUS 'VICE' GRIP:
The classical or 'claw' grip (above left), also known as the C-E technique, can be used with either a one or two handed approach to face mask ventilation. When performed one-handed in the presence of a soft collapsible reservoir bag it has the advantage of providing the airway operator with the opportunity to use the other hand to get a 'feel' for lung compliance as well as the rapid identification of primary leaks as outlined above. It is limited, however, by the fact that jaw thrust can only be applied by the small fifth finger.
The 'vice grip' (below left), also known as the V-E technique can only be performed using both hands. It involves placing the thumbs and thenar eminences longitudinally along each side of the mask to create a seal. The second to fifth digits are then oriented almost vertically such that they are all able to reach behind the angle of the mandible and participate in lifting the mandible towards the mask. This simultaneously produces strong jaw thrust, lifting the tongue off the posterior pharynx to improve patency as well as holding the mask and face in the 'vice' created by the thumb and fingers, augmenting the seal . Using this grip it is even possible to lift the head into a more pronounced 'sniffing position' to further improve airway patency.
The Vortex Approach strongly advocates that a best effort at face mask ventilation should always include vice grip.
As the vice grip technique is easier to master/retain it is recommended for routine first line use by clinicians who infrequently perform face mask ventilation. For all clinicians, alternating between the two grips can be a useful strategy to avoid fatigue during prolonged episodes of face mask ventilation.

Supraglottic Airway
BEST EFFORT
Supraglottic Airway
BEST EFFORT
troubleshooting supraglottic Airway INsertion
As with the other lifelines, interventions to optimise attempts at achieving alveolar oxygen delivery via a supraglottic airway should be targeted towards the factors impeding success. The challenges arising with supraglottic airway insertion can be categorised under three headings:
- Entry: inability to insert the supraglottic airway into the mouth due to factors such as limited mouth opening or excessive muscle tone.
- Passage: difficulties in getting the supraglottic airway to negotiate the pathway through the pharynx to reach the larynx.
- Seating: misalignment of the laryngeal orifice and the laryngeal inlet (leading to obstruction or gastric insufflation) or inadequate seal of the cuff of the supraglottic around the laryngeal orifice (leading to leak around the cuff and inability to generate sufficient pressure to allow alveolar ventilation).
best effort at supraglottic airway
The process of optimisation for supraglottic airway differs somewhat from that of the other lifelines. Sequential attempts to establish alveolar oxygen delivery using a endotracheal tube or face mask typically involve optimisation techniques being superimposed on one another producing incremental gains to improve the view of the larynx during intubation or the patency of the airway during face mask ventilation. Whilst such escalating optimisations targeted towards the specific issues impeding alveolar oxygen delivery also occur when using a supraglottic airway, a significant number of optimisations targeted at passage of a supraglottic airway involve alternative rather than incremental strategies. The significance of this is twofold:
- Firstly it means that previous attempts at passage of a supraglottic airway may provide limited information about the likelihood of success at subsequent optimised attempts (beyond identifying the nature of the problem - entry, passage or seating - and ensuring interventions are selected that will impact on this issue). For example, if a supraglottic airway 'holds up' trying to negotiate the turn at the posterior oropharynx into the laryngopharynx when passed conventionally, beyond an awareness that they are frequently useful techniques to overcome this problem in general, no information is usually gained that would allow the airway clinician to predict the likely impact of using a different type of supraglottic airway or inserting the supraglottic airway backwards in that particular patient. This is different to the situation during laryngoscopy in which the view obtained with one laryngoscope blade may provide important information about the likely impact of using an alternative blade. This informs the decision about the best intervention to further optimise the airway on a subsequent attempt and in some situations may help decide whether further attempts are warranted at all.
- Secondly it means that there are key optimisations for supraglottic airway that cannot be implemented simultaneously during the same attempt but instead necessitate separate attempts. For example, although optimisations such as positioning and muscle relaxation can (and should) be superimposed during a single attempt, the introduction of a supraglottic airway using both conventional and reversed orientation clearly requires two separate attempts.
“when a supraglottic airway is used as a rescue device...the first attempt...should be made with the device with which the clinician feels most confident they will be able to achieve alveolar oxygen delivery”
The above two factors conspire to make it more difficult to achieve a best effort at supraglottic airway insertion in under three attempts, particularly in the situation in elective anaesthesia where a supraglottic airway is the intended primary airway and for surgical reasons a type, other than that with which the airway operator is most confident, is used on the first attempt. Thus taking the example of induction of anaesthesia for oral surgery: if a reinforced supraglottic airway was used on the first attempt but could not be passed around the back of the oropharynx, attempts with a standard supraglottic airway using both conventional and reversed orientation of the device would typically be reasonable before declaring a best effort. Whilst other optimisation interventions such as positioning and muscle relaxation should also be superimposed during these successive attempts, the above example highlights that even with the most efficient implementation of all other optimisations, the high impact 'size/type' and 'manipulation of device' interventions that would be employed in this situation will in themselves require three attempts to implement. The result is that clinicians must be particularly aware of the need to rapidly escalate optimisations in order to achieve a best effort at supraglottic airway within the three attempt limit. This may be less of an issue when a supraglottic airway is used as a rescue device than as the intended primary airway, as in this situation the first attempt with supraglottic airway should made with the device with which the airway clinician feels most confident they will be able to achieve alveolar oxygen delivery.
The following table outlines the possible real-time optimisation interventions in pursuit of a best effort at supraglottic airway and the issues they target. Note that this table is intended as a foundation resource for training and not to be used during the process of airway management.
Note: performance of flextension (sniffing position) must only be undertaken following consideration of the need for cervical spine precautions.
Use of a Laryngoscope to Optimise Supraglottic Airway Placement:
Inability to pass a supraglottic airway when entry of the device into the mouth is possible, typically results from the cuff of the device folding over on itself or from it pushing the tongue or epiglottis back and obstructing the airway. The use of a laryngoscope to assist with placement of a supraglottic airway is often not considered, but is able to overcome most of the issues associated with difficult passage by lifting the tongue and epiglottis forwards and creating a clear space into which the supraglottic airway can be placed - even if a view of the larynx cannot be obtained. Due to the high profile of the laryngoscope there is often limited space to insert the supraglottic airway in the midline but with practice (in non-emergency situations) it is usually easy to achieve entry via the left corner of the mouth. This is easier when a device without a fixed curve in the stem is used (e.g. classic or proseal LMA or iGel). A useful strategy is to leave the laryngoscope in situ following the last attempt at endotracheal tube and immediately attempt placement of the supraglottic airway with laryngoscope assistance. A device known as a 'tongue anterioriser' is also available that performs a similar function to the laryngoscope but without providing any view into the pharynx (which is not required for this technique). The advantage of the tongue anterioriser is that it is flat, this lower profile making it less likely that it will will obstruct entry of the SGA into the mouth.
Use of a Bougie to Optimise Supraglottic Airway Placement:
As a rescue technique, the deliberate passage of a bougie into the oesophagus followed by railroading of a second generation supraglottic airway over the bougie via it's oesophageal lumen, is well described. The technique is mentioned in the 2015 Difficult Airway Society Guidelines where it is described as having a 100% success rates on the first attempt (superior to success rates using digital insertion or insertion using the Proseal introducer) when performed using a Proseal LMA in some studies. The rationale is that the bougie guides the oesophageal opening on the tip of the SGA to the oesophagus, which in turn better aligns the oesophageal lumen with the laryngeal inlet. This technique is likely to be more difficult with some designs of LMA and so requires practice with the device being used in a non-emergency situation. As there is a risk of trauma from oesophageal passage of the bougie it should only be attempted as a rescue technique. This technique can be combined with the use of a laryngoscope as described above, passing first the bougie, then the supraglottic airway with the laryngoscope in situ.

Endotracheal Tube
BEST EFFORT
Endotracheal Tube
BEST EFFORT
trouble shooting endotracheal intubation
The process of endotracheal intubation can be divided into three components:
View: getting a view of the larynx
Delivery: delivering the endotracheal tube to the laryngeal inlet
Passage: feeding the endotracheal tube from the laryngeal inlet into the trachea
Intubation may become challenging due to the inability to complete one or more of these steps and optimisation interventions should be targeted accordingly.
BEST EFFORT at endotracheal tube
The following table outlines the potential real-time optimisation interventions in pursuit of a best effort at endotracheal intubation and the issues they target. Note that this table is intended as a foundation resource for training and not to be used during the process of airway management.
Note: performance of flextension (sniffing position) must only be undertaken following consideration of the need for cervical spine precautions.
Optimising success when using a Hyperangulated Videolaryngoscope:
While a hyperangulated videolaryngoscope will often provide a good view of the larynx with minimal skill requirement, familiarity with the device is required to recognise the view required to facilitate passage of the tube. Instinctively clinicians will aim to achieve a close up view of the laryngeal structures but this view often leads to the laryngoscope itself interfering with passage of the endotracheal tube. Thus while hyperangulated videolaryngoscopy removes much of the skill required in getting a view of the vocal cords, considerable skill is still required to achieve delivery of the tube to the larynx and its subsequent passage into the trachea. It is useful to think of the techniques/devices required to achieve each of the components of intubation when using a hyperangulated video laryngoscope.
View: the hyperangulated videolaryngoscope is excellent at achieving this via an indirect view but (in contrast to a conventional geometry laryngoscope) its design does not provide any opportunity for getting a direct view of the larynx, as the extreme curvature of the blade prevents it displacing the tongue to achieve this.
Delivery: the inability to achieve a direct line-of-sight when using a hyperangulated videolaryngoscope means that a direct line-of-delivery of the ETT to the cords is also impossible. Thus the hyperangulated videolaryngoscope is not recommended as a first line device unless a getting a direct view of the cords is known to be impossible or judged to be extremely unlikely, as it may complicate tube delivery despite a good laryngeal view (even compared with when a poor direct view is possible). If a direct view of the larynx has proven impossible, however, on the first attempt at intubation a hyperangulated videolaryngoscope is the recommended blade for a second attempt, if the device and an appropriately skilled airway operator are available. This is consistent with the approach advocated in the 2018 UK National Guidelines for management of tracheal intubation in critically ill adults. Optimising delivery of the ETT to the larynx requires:
Appropriate view: a 'wide-angle' rather than a 'close-up' view of the larynx makes it easier to identify where the ETT/introducer is coming from as well as providing more space for introduction of the tube by preventing the laryngoscope blade from 'crowding' the larynx. In addition, obtaining only a view of only the posterior half of the vocal cords (grade 2 view) rather than their entirety (grade 1 view) assists with bringing the axis of the view provided by the hyperangulated blade and that at which the bougie is introduced, in line with the long axis of the trachea.
Curved introducing device (stylet/bougie): this is mandatory when using a hyperangulated blade to navigate its curve and prevent the tube from persistently going posterior to the laryngeal inlet. Use of a bougie/stylet with a steerable tip can further facilitate delivery. A fiberoptic scope (either without or with the screen attached to it - the latter being referred to as fiberoptic assisted video intubation or FAVI) can also be used as a steerable introducing device in this situation.
Passage: as the hyperangulated blade (in combination with the patient anatomy which made direct laryngoscopy difficult in the first place) results in the stylet/bougie approaching the larynx at a steep angle from behind, it will often hit the anterior wall of the trachea once it passes through the laryngeal inlet, preventing the bougie or ETT from being fed into the trachea. Again the use of a stylet/bougie with a steerable tip can facilitate passage of the bougie/ETT into the trachea as flexing the tip allows it to be brought into alignment with the long axis of the trachea once it has traversed the laryngeal inlet. Passage of the ETT into the trachea may also be impeded by 'hang up' the ETT tip on the arytenoids due its posterior approach to the larynx. Standard manoeuvres to overcome this such as rotating the ETT 90 degrees anticlockwise may be impeded by the fixed curve on the introducer device. Use of a 'bullet tipped' ETT (& possibly a smaller size) can help overcome this.
Thus in a time-critical situation, to achieve intubation as rapidly as possible when a direct view has proven impossible, it is useful to consider the empirical use of the following three devices together as a 'package deal':
Hyperangulated Videolaryngoscope: to facilitate a view.
Curved steerable tip bougie/stylet: to facilitate delivery.
Bullet tipped ETT: to facilitate passage.